Light-Powered Catalyst Breakthrough: A New Era in Chiral Molecule Synthesis
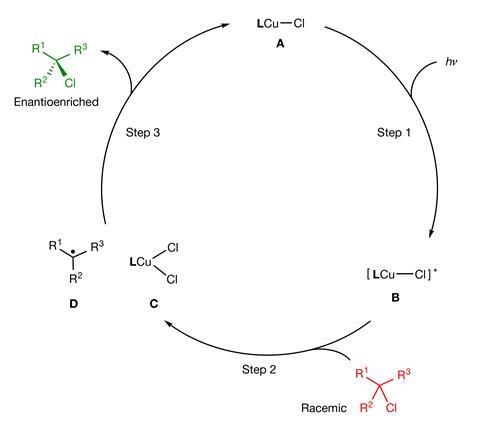
In a groundbreaking advance in the world of materials science and photochemistry, a team of researchers led by Gregory Fu (Caltech) and Peng Liu (University of Pittsburgh) have developed a light-activated catalyst that resets carbon–halogen bonds—paving the way for the efficient synthesis of single-enantiomer compounds. This novel strategy transforms racemic mixtures into highly enantioenriched products, solving a long-standing challenge in the synthesis of chiral pharmaceuticals.
Why This Matters
Chirality—the "handedness" of molecules—is a critical factor in pharmaceutical activity. Producing only the biologically active enantiomer can be the difference between therapeutic efficacy and harmful side effects. Traditional approaches to deracemization have struggled with alkyl halides due to the strength of their carbon–halogen bonds. But this new technique, published in Nature, marks the first time photochemistry has successfully targeted bonds involving heteroatoms.
The Science Behind the Spark
The catalyst employs a copper chloride complex attached to a chiral phosphine ligand. Upon light activation, it initiates a single-electron transfer reaction with an alkyl halide, cleaving the bond and generating a radical. This is then recombined with a chloride ion from the catalyst, re-forming the carbon–halogen bond but with precise stereochemical control.
“Others have used photochemistry for carbon–carbon or carbon–hydrogen bonds,” notes Fu. “But this is the first time we’ve seen this kind of control over carbon–halogen bonds via light.” The reaction’s elegance lies in its ability to manipulate such stable bonds without the need for extreme conditions.
Implications for Pharmaceuticals and Beyond
According to Peng Liu, the new approach offers a generalizable route to many types of carbon–heteroatom bonds, vastly expanding the chemical space for drug development. Eric Ferreira (University of Georgia), who reviewed the study, praised it as “remarkable” and believes this concept could be extended to new classes of molecules with slight catalyst modifications.
What’s Next?
With the foundations laid for broad substrate scope and scalability, this technology may soon be a cornerstone of sustainable pharmaceutical manufacturing. By integrating computational modeling with experimental chemistry, the team demonstrates how quantum chemistry and light-driven processes can unlock new levels of precision in synthetic design.

Comments
Post a Comment